Relenza flu treatment
1999
drug that prevents the spread of influenza virus
This drug promises to revolutionise the treatment of influenza, a viral disease that kills many thousands of people every year and causes massive discomfort and loss of productivity for countless others.
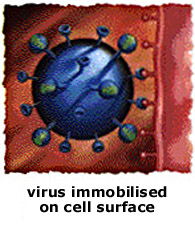
In the 1970s Dr Graeme Laver at the Australian National University isolated and accidentally crystallised the protein sialidase, one of two proteins in the outside coat of every influenza virus. In 1978 he joined Dr Peter Colman of the CSIRO's Division of Biotechnology to study the crystal. Using a technique called X-ray crystallography, they magnified the protein molecule 100 million times to reveal individual atoms. Now that they could 'see part of the enemy', they studied how the protein changed with each new generation of flu virus.
In 1982 they identified a part of the molecule that always stayed the same! This is important because the virus mutates every year to become slightly different, requiring a new vaccine to be developed each year.
In 1986 Professor Mark von Itzstein at Monash University began searching for a chemical that would plug into the place on the molecule that stayed the same. This would lock the flu virus onto the cell it was attacking and stop it from reproducing and spreading.
But the CSIRO and universities couldn't afford to fund development. Australian company Biota Holdings agreed to purchase the patents and fund development of the drug at the Victorian College of Pharmacy.
In 1989 Biota licensed Glaxo Australia to manufacture and conduct trials on the drug.
Laboratory trials on ferrets were spectacularly successful. Ferrets are particularly susceptible to human flu. In 1993 the USA's Food and Drug Administration gave permission for the drug to be tested on humans. In 1999 the flu drug, called Relenza and manufactured by Glaxo Wellcome, was approved for use in Australia, USA and Europe. By the end of the century it was available in over 50 countries including Japan.
In 1996, Laver, Colman and von Itzstein shared the Australia Prize for their work.
Who Did It?
Key Organisations
Biota Holdings Limited : R&D, funding
CSIRO Division of Protein Chemistry : research
Glaxo Australia : manufacture, testing
Key People
Dr Peter Colman : CSIRO X-ray crystallography
Dr Graeme Laver : ANU researcher
Prof Mark von Itzstein : Monash University drug research
Further Reading
'US clinical trials for flu vaccine'
Jane Ford (ed)
Scitech, December 1993.
Links
CSIRO
Australia Advances. Quicktime video about Relenza
CSIRO
Disarming
flu viruses. Scientific American January 1999
Glaxo Wellcome Relenza page
Biota Holdings Ltd
British
National Health Service view on Relenza
US
Food and Drug Administration Relenza page
Australian
Academy of Science. An end to the flu?
|